By Jenelle Patterson
We’d all like to slow the hands of time and stay young forever, and scientists have been trying for centuries to figure out what causes aging. Research into human aging has now identified some causes, but can this information be applied to horticultural crops? Here, we will explore how these two fields synergize and how combining them can advance plant research.
UNDERSTANDING METABOLISM
Before diving into why
cells die, we need to understand how cells live. The majority of the living
that goes on in cells is done by enzymes. Simply put, enzymes are proteins that
have a job. For example, the enzyme amylase converts starch in our mouths and
intestines into simple sugars that are absorbed and used for energy. The term
metabolism describes the network of enzymes and chemical reactions that allow
cells to live and function.
Metabolism includes both primary metabolism, which controls the reactions required for life, and secondary metabolism, which involves the production of molecules that make life more interesting (think flavor and color molecules in tomatoes). Typically, metabolism works like a well-oiled machine but sometimes makes mistakes that produce damaged molecules.
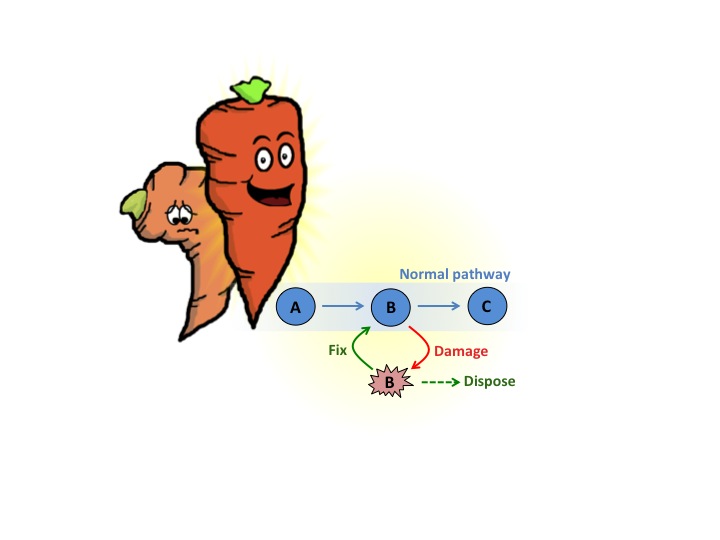
Why should we care about a few damaged molecules if metabolism gets it right most of the time? Unfortunately, metabolite damage can negatively impact plant performance in several ways. First, damaged molecules are simply trash that must be disposed of, and this is a wasteful process that can rob yield. Second, and even worse, some damaged molecules are outright poisonous and, in the long term, can even kill the plant that makes them.
Scientists have identified ways that human and bacterial cells deal with damaged molecules. The cells produce enzymes that prevent the buildup of damaged molecules either by repairing them (recycling them to their original undamaged form) or disposing of them by turning them into something harmless. In other words, the enzymes fix the damage or safely trash the damaged products.
In humans, recycling of damaged products provides nutrients to maintain cellular function and eliminates harmful waste. Rates of damage repair decline during aging, which has been linked to the onset of devastating age-related diseases such as cancer, heart disease, diabetes and neurodegeneration. Exciting new medical technologies are being developed to increase healthy lifespan by targeting the buildup of damaged molecules using pharmaceuticals and gene therapy.
APPLYING THE SCIENCE TO PLANTS
Luckily, humans and
plants aren’t all that different. By some estimates, we are genetically
approximately 50 percent identical to plants! Since a lot of human and plant
metabolism is similar (e.g., we both use sugar to make energy molecules), it
stands to reason that the ways in which molecules can be damaged and repaired
could also be similar. The genetic similarity between humans and plants allows
researchers to use the vast information about the mechanisms that prevent
disease to identify enzymes with similar functions in plants (a process called
“comparative genomics”).
Plants obviously don’t develop neurological disorders when aging, but the accumulation of damaged molecules can kill cells and their repair uses precious energy. The combination of cell death and energy starvation decreases the length of time that fruits and vegetables retain quality after harvest. Engineering or breeding plants to be more efficient at damage repair, or decreasing damage in the first place, could make the energy saved available for desirable traits like enhanced yield.
Increasing yield is the most obvious goal of applying damage-repair research in plants, but there are also more creative uses. For example, there are current efforts to engineer plants to improve color, flavor or nutrient content, or even to make molecules that plants don’t naturally produce (e.g., antibodies). Since we don’t know how tinkering with these processes will affect metabolism as a whole (since it is an interwoven network), it is entirely possible that damaged molecules will be produced as unintended byproducts. Thus, discovery of damage-repair enzymes may be crucial for the advancement of this type of biotechnology.
So, will the application of our knowledge about human aging lead us to produce everlasting strawberries or tomatoes? Probably not. But, by using what we learn from comparative genomics and damaged molecule repair, we might see longer-lasting and more nutritious fruits and vegetables in the future.
Jenelle Patterson is a postdoctoral associate at the University of Florida.
This article was featured in the January issue of VSCNews magazine. If you would like to receive future issues of VSCNews magazine, click here.
Share this Post